1 min read
Guide to a Faster Engineering Change Order Procedure in Electronics
Change management is usually a requirement in the electronics industry. This specifically relates to changes needed after the initial design of your...
2 min read
 Matric Group
:
Updated on March 20, 2026
Matric Group
:
Updated on March 20, 2026


Electronic contract manufacturers (ECMs) regularly tout the certifications they possess -- just take a look at their websites. Hey, even we do it. And it’s for good reason: Certifications communicate what your company is capable of to potential customers. It also communicates trustworthiness and dependability. If you’re a medical company seeking electronics manufacturing, the ISO 13485 certification is the most important one to you.
This certification is for quality management of medical electronics, an offshoot of the more general ISO 9001 certification. When your company reaches out to potential new ECMs, the phrase ISO 13485 is likely uttered from one of the parties within the first few sentences – but what about this certification makes it important?
Some OEMs require their suppliers to be certified in ISO 13485. Having a customer certified in ISO 13485 doesn’t necessarily mean they’re a better option than another electronics manufacturer that doesn’t possess the certification. But it does means that they are proven to abide by standards set forth by the FDA’s Quality System Regulation and Current Good Manufacturing Practices.
ISO 13485 is audited by a third party who reviews the ECM’s ability to meet regulatory and customer requirements and maintain effective processes and risk management. In essence, it’s a badge of approval that communicates authenticity to an OEM.
ISO 13485 isn’t the only quality management system certification. ISO 9001 is similar but focuses more on business aspects not found in 13485. Therefore, some contract manufacturers choose to secure both certifications to better represent the company infrastructure. The key differences between the two are:
When a manufacturer wishes to obtain a 13485 certification, the first step is to learn about the certification is and its requirements. Once comfortable with the standards of the management system’s requirements, the manufacturer must implement them into its organization.
This all starts with a quality manual. This document outlines the quality management system’s:
This can be outsourced or done within your vendor’s quality department. Then a gap analysis is performed to identify requirements that are missing within your vendor’s existing system. Once the manual and procedures are written and the system is put into motion, its time for to review.
The contract manufacturer needs to schedule a time with an accredited auditor to visit its facility and review the system firsthand. The manufacturer has to keep the location of customers like you in mind. For example, a company wanting to do business with European customers should make sure it meets EU standards. Some countries don’t recognize registration from foreign auditing bodies, so watch who you work with.
The review itself will have multiple phases.
Your ISO-certified vendor has to undergo yearly audits to maintain it. Electronics manufacturers are OK with this tedious process because it proves they have proper systems in place to satisfy your medical electronics needs.
Matric and Dynamic Manufacturing have both ISO 13485 and ISO 9001 certifications. Let us know if you have any questions about either crucial certification.

1 min read
Change management is usually a requirement in the electronics industry. This specifically relates to changes needed after the initial design of your...

1 min read
Without a doubt, the development of enhanced inspection equipment has been critical to the PCB assembly industry. The speed and precision of...
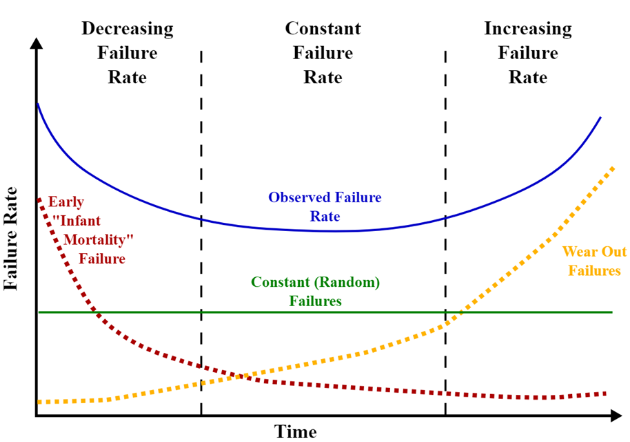
1 min read
Testing and inspection are vital parts of electronics contract manufacturing, for obvious reasons. Before that shipment of PCBs arrives on your...